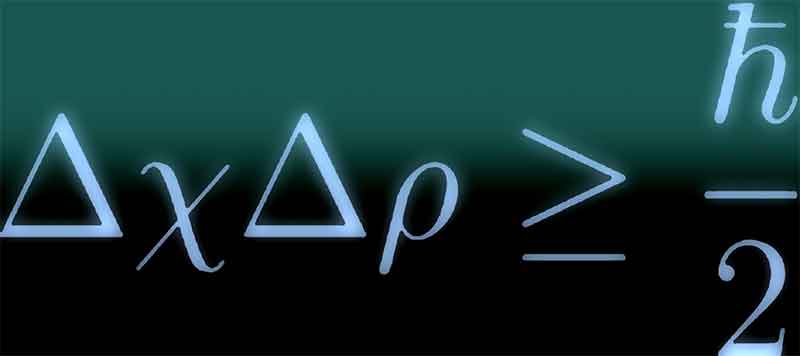 
That is, light isn’t a particle or a wave-it has properties of both. Thanks to a pioneering test known as Young’s Double-Slit experiment, physicists discovered the answer was “Door No. The origin of the uncertainty principle is found in the duality of particles in quantum physics depending on what they’re doing, they can be described as either a particle or a wave, Orzel tells Popular Mechanics.Īt the turn of the 20th century, physicists were engaged in a heated debate regarding the nature of light, and whether it exists as a particle or a wave. In his book, How to Teach Quantum Physics to Your Dog, he covers Heisenberg’s uncertainty principle. Getty Images What Is the Heisenberg Uncertainty Principle?Ĭhad Orzel is an associate professor in the Department of Physics and Astronomy at Union College in Schenectady, New York, who is also the author of several books that explain often complicated and esoteric ideas to a layman audience. Reality is telling us that we can have our quantum cake, but we can’t eat it, too. Heisenberg recognized that if it were possible to measure both the momentum and the position simultaneously with greater accuracy, quantum mechanics would collapse. Legendary physicist and master bongo player Richard Feynman put it like this: “The uncertainty principle ‘protects’ quantum mechanics. It doesn’t matter how smart you are, or how sophisticated your equipment, is you can’t think your way past it. It’s special because it remains intact no matter how good our experimental methods get this isn’t a lack of precision in measurement. German theoretical physicist Werner Heisenberg first introduced his uncertainty principle in a 1925 paper. If this was all Heisenberg’s uncertainty principle said, it probably wouldn’t have been profound enough to weave its way into pop culture in the form of mugs, T-shirts, and cartoons-let alone place its pioneer as an alias for an infamous meth-cooking chemistry teacher. But every physics graduate first starts to unpack this concept through the lens of position and momentum, so we will, too. The principle also applies to other pairs of characteristics in quantum systems, like energy and time. 
In its most basic and commonly known form, the uncertainty principle says that the more precisely you know the position of a particle in a quantum system, the less well you know its momentum (and vice-versa). Whether you’re laughing right now-or staring at your screen in confusion-hinges on how much you know about one of the foundational ideas in quantum physics: Heisenberg’s uncertainty principle.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
